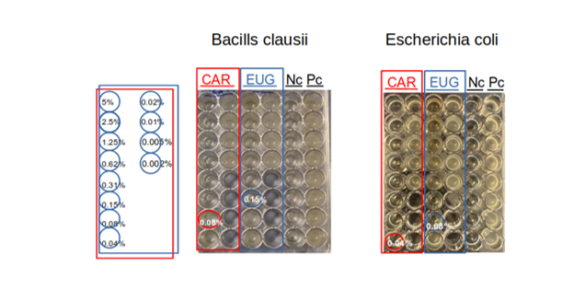
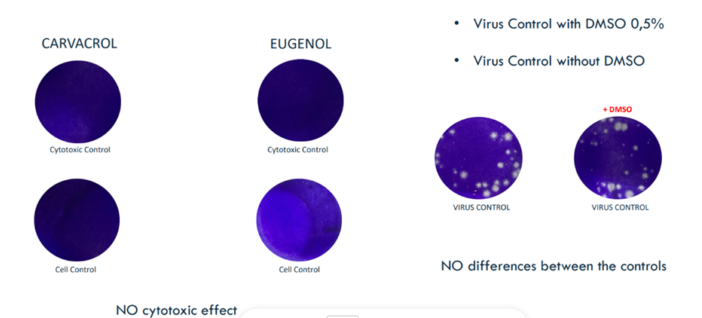
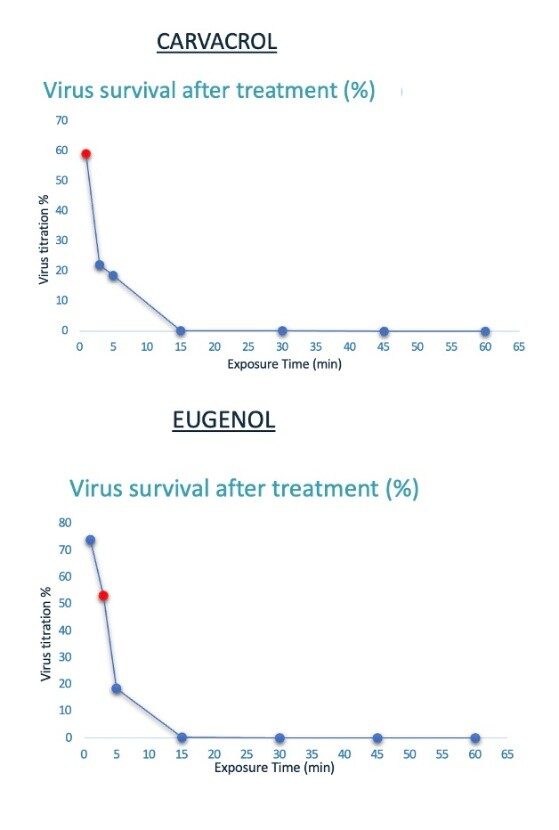
RELIANCE project presented at LOPEC 2023
RELIANCE was presented at LOPEC 2023 Conference that was recently held in Munich, Germany, attended by experts from 22 countries who shared their expertise in around 170 presentations. The paper-based electrochemical (bio)sensors that will be used as an analytical method in the project were presented in the session dedicated on “Challenges and use cases of large area, printed or organic electronics in biomedical applications which included all applications in biomedical and healthcare such as sensors, diagnostic devices and wearable health patches that contain significant printed and flexible elements”.
“As the world’s most important communication platform for the industry, the LOPEC Conference presented the latest state of the technology and provided valuable insights for both new users and industry insiders,” said Wolfgang Mildner, General Chair of LOPEC.
In various conference formats, companies such as BMW, Microsoft, Pirelli, Samsung, Sun Chemical and Varta presented trends and new applications in printed electronics.
“The LOPEC Conference is a great yearly event for me to bring myself up to date,” stated Dr. Edzer Huitema, Chief Technology Officer at E Ink.
Focus topics of the exhibition and conference were Smart Living and Mobility, as in the words of Mildner, “sustainability has come into even sharper focus, because printed electronics are produced in a resource-efficient manner and enable green solutions”.